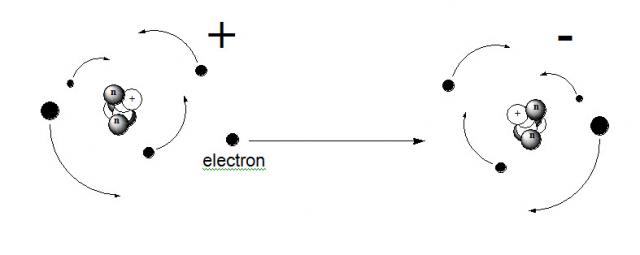
Formation of Positive IonsPositively charged ions are called cations. Cations form when an atom loses one or more of its valence electrons. It does this when it needs to attain a noble gas configuration.
Take Potassium and Argon for example. The electronic configuration for Potassium is: 1s2 2s2 1p6 3s2 2p6 4s1. While Argon's configuration is: 1s2 2s2 1p6 3s2 2p6. Potassium has one more electron (4s1). If Potassium loses this electron, its configuration will be the same of Argon. This will make Potassium's charge to be positive. Although Potassium and Argon would have the same configuration, this would not change the Potassium atom into an Argon atom. |